If The Earth is an Enclosed System, Why Are There Atmospheric Pressure Gradients?
The atmospheric pressure gradients above the Earth are the result of a combination of factors, including varying Sun induced temperatures, types of gases, altitude, molecular densities, molar mass, molecular compositions, and atomic bonding indexes, which are all contained within The Earth’s atmospheric layering system.
Gasses separate in accordance to these aforementioned variables in any enclosed system.
Analysis
Gases that are lighter than air include water vapor, methane, hot air, hydrogen, neon, nitrogen, ammonia, and helium. These gases have a lower density than air, which causes them to rise and float in the enclosed pressure system of The Earth’s atmosphere.
Simply put, you first must have gas pressure, which is caused by a container, and then theses gaseous gradients separate within this container, according to temperature, pressure, and elemental composition (electron density). For instance, oxygen is a heavier gas than hydrogen, and so they “gradiate” at different altitudes, and with varying distributions. Add The Sun’s effect on temperature differentials, and this additionally effects the separation of atmospheric layers.
Solar Radiation Affecting The Earth’s Atmosphere
Solar radiation, often called the solar resource or just sunlight, is a general term for the electromagnetic radiation emitted by the Sun. In Earth's atmosphere, the Sun has the largest impact upon the Stratosphere, which is where the ozone layer is. The Troposphere is the layer below the Stratosphere, which is where weather occurs. Heating up the lower Stratosphere heats up the upper Troposphere. If the surface of the Earth is hot, and the troposphere is cold, then the temperature difference causes strong updrafts. Thereby effecting the overall atmospheric pressure distribution.
Hydrogen
Hydrogen is the simplest element. Each atom of hydrogen has only one proton. Hydrogen is the lightest element. Hydrogen is a gas at normal temperature and pressure, but hydrogen condenses to a liquid at minus 423 degrees Fahrenheit (minus 253 degrees Celsius). Hydrogen bonds can be intermolecular (occurring between separate molecules) or intramolecular (occurring among parts of the same molecule). The energy of a hydrogen bond depends on the geometry, the environment, and the nature of the specific donor and acceptor atoms and can vary between 1 and 40 kcal/mol. Because of Hydrogen’s light composition, it will interact with atomic systems with a different distribution than something as heavy as Argon.
Argon
Argon is also one of the heaviest gasses known to Man, with an atomic weight of 39.948 u. Argon is a chemical element with the symbol Ar and atomic number 18. It is in group 18 of the periodic table and is a noble gas. Argon is the third-most abundant gas in Earth's atmosphere, at 0.934% (9340 ppmv). It is more than twice as abundant as water vapor (which averages about 4000 ppmv, but varies greatly), 23 times as abundant as carbon dioxide (400 ppmv), and more than 500 times as abundant as neon (18 ppmv).
Nitrogen
Nitrogen is one of the most abundant gases in the Earth’s atmosphere, making up 78 percent of its volume. It is also one of the heaviest gases, with a molar mass of 28.02 grams per mole. Oxygen makes up 21 percent of the Earth’s atmosphere and has a molar mass of 32 grams per mole.
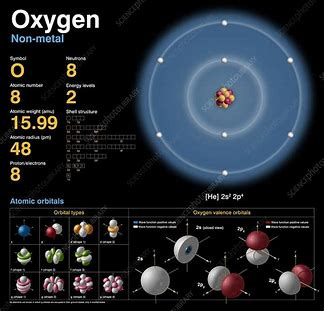
Oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as well as with other compounds. Oxygen is Earth's most abundant element, and after hydrogen and helium. At standard temperature and pressure, two atoms of the element bind to form dioxygen, a colorless and odorless diatomic gas with the formula O squared.
Water vapor is present in very small amounts in the atmosphere but still contributes to its overall weight. It has a molar mass of 18 grams per mole. Carbon dioxide makes up 0.04 percent of the Earth’s atmosphere but is still considered one of its heavier gases because it has a molar mass of 44 grams per mole.
Again, in simple terms, the weights of these gases vary depending on their temperature and pressure. For example, at standard temperature and pressure (STP), nitrogen has a density of 1.2506 grams per liter while oxygen has a density of 1.4294 grams per liter . However, at higher temperatures or pressures, these densities will change slightly, as The Sun’s rays affect them.
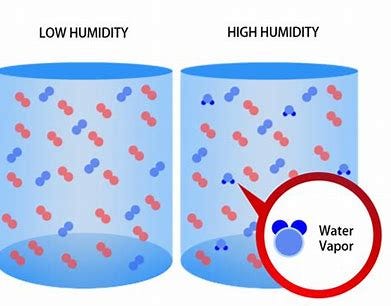
Additionally, other factors such as humidity can also affect gas densities. There are several heavy gases found in our atmosphere but their weights can vary depending on conditions. Nitrogen, oxygen, water vapor, and carbon dioxide are all considered to be heavy atmospheric gases. Humidity is the concentration of water vapor present in the air. Water vapor, the gaseous state of water, is generally invisible to the human eye. Humidity indicates the likelihood for precipitation, dew, or fog to be present.
The most abundant molecules in the Exosphere are atomic oxygen, helium, and hydrogen. A large part of the atomic hydrogen in the Exosphere originated from water in the lower atmosphere, rather than from molecular hydrogen, which gain, is affected by molecular density, molecular composition, atomic bonding, and temperature as is dictated by solar radiation.
The Earth’s Atmospheric Composition: Nitrogen, Oxygen, Argon and CO2, and Traces Gases:
1. Nitrogen (78.1%)
2. Oxygen (20.9%)
3. Argon (0.93%)
4. Carbon Dioxide (0.04%)
5. Trace Gases
Adding varying Sun driven temperatures to these elements, these atmospheric gasses separate into layers above the Earth in its enclosed system.
Additionally, the equilibrium pressure of a gas, just like the equilibrium temperature of a gas, is a macroscopic property applicable to the collection of gas molecules inside a container, not a microscopic property applicable to individual gas molecules at every point inside the container. Considering the walls of the container, while the impact forces of individual molecules on the walls of the container will vary, it is the average of the impact forces of a collection of molecules that determines the macroscopic property of pressure. Similarly it is the average kinetic energy of the molecules that determines the macroscopic property of the temperature of a gas, not the kinetic energies of the individual molecules which will vary above and below the average.
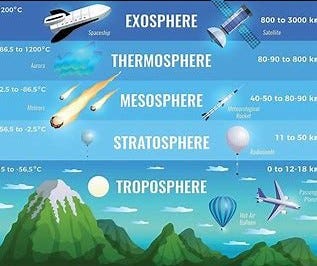
The Layers Within Earth's Atmosphere Include:
Troposphere
The Troposphere starts at the Earth's surface and extends 8 to 14.5 kilometers high (5 to 9 miles). This part of the atmosphere is the most dense. Almost all weather is in this region.
Stratosphere
The Stratosphere starts just above the troposphere and extends to 50 kilometers (31 miles) high. The ozone layer, which absorbs and scatters the solar ultraviolet radiation, is in this layer.
Mesosphere
The Mesosphere starts just above the stratosphere and extends to 85 kilometers (53 miles) high.
Thermosphere
The Thermosphere starts just above the mesosphere and extends to 600 kilometers (372 miles) high.
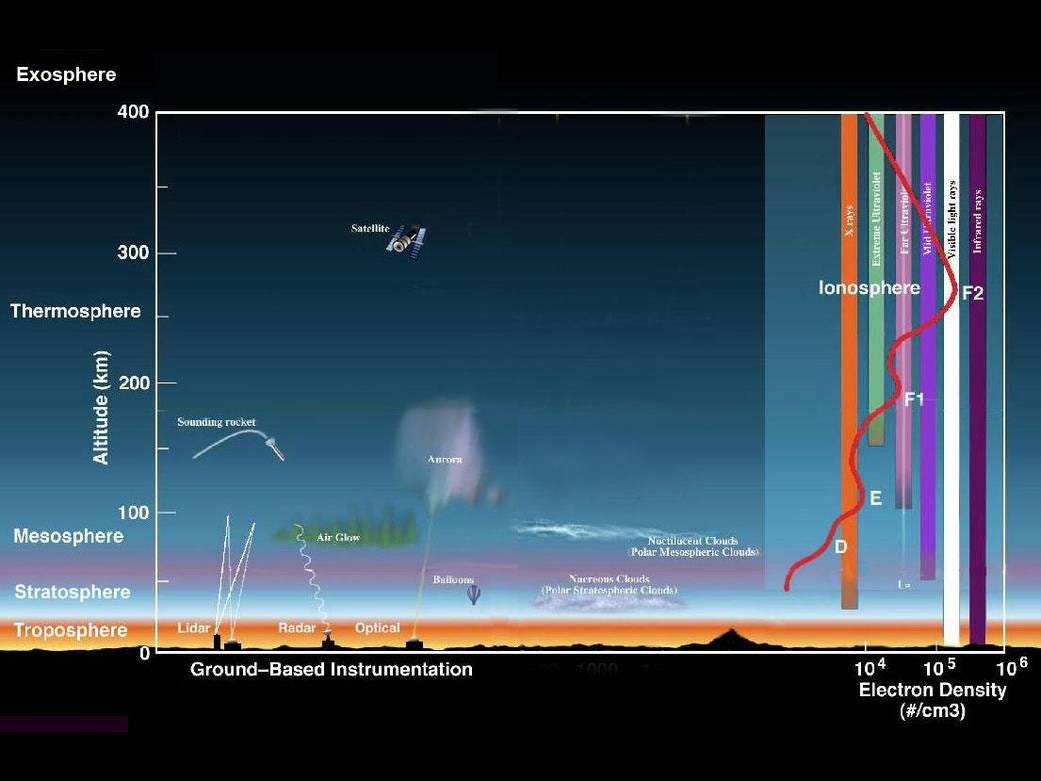
Ionosphere
The Ionosphere is an abundant layer of electrons and ionized atoms and molecules that stretches from about 48 kilometers (30 miles) above the surface to the edge of space at about 965 km (600 mi), overlapping into the Mesosphere and Thermosphere. This dynamic region grows and shrinks based on solar conditions and divides further into the sub-regions: D, E and F; based on what wavelength of solar radiation is absorbed.
Exosphere
This is the upper limit of our atmosphere. It extends from the top of the thermosphere up to 10,000 km (6,200 mi).
--Credit: NASA/Goddard
Conclusion
Put simply, atmospheric pressure gradients in The Earth’s enclosed Thermodynamic system are the result of the interplay between various factors, including altitude, molar mass, molecular density, molecular concentration. molecular composition, humidity, atomic bonding indexes, and temperature, as dictated by solar radiation. Without containment, and if contiguous to vacuum, these variables would dramatically change, resulting in the complete destruction of The Earth’s atmospheric layering system as all these layers disperse out into the vacuum Void of Outer Space.
Can Gravitational Attraction Hold The Earth’s Atmospheric System Intact Contiguous to the Vacuum of Outer Space?
Lastly, In The Heliocentric Model, Newton’s Law of Gravitational Attraction applies to all objects. However there’s a caveat. At the smaller levels of mass, the gravitation attraction effect is so small it is negligible, meaning it so small or unimportant as to be not worth considering. It is mathematically and observably insignificant. According to Newton’s Law of Gravitation, the gravitational force on a body is proportional to its mass.
Note:
In Newtonian Physics, BOTH masses under scrutiny must contain a large mass in order to measure and observe gravitational attraction, Both The Earth, and the mass it is in relationship to, must contain a large mass in order to experience the conditions necessary for significant gravitational attraction. Despite The Earth’s larger mass, when compared to single atom at the highest elevations above The Earth, there is insufficient mass in the higher altitude atom to establish any significant gravitational attraction between The Earth and that atom because, as Newton’s Law of Gravitation states, the gravitational force on a body is proportional to its mass.